Original Article - DOI:10.33594/000000604
Accepted 22 January 2023 - Published online 07 February 2023
Links Between Vitamin D-Binding Protein, Alpha-1 Antitrypsin and Neutrophil Proteins in Meconium
Keywords
Abstract
Background/Aims:
Alpha-1 antitrypsin (AAT), vitamin-D binding protein (VDBP) and neutrophil granule proteins are specifically related to the neutrophil function and may be considered candidate biomarkers detected and measured in meconium (the first feces of newborn infants) as signals indicating abnormal processes in the fetal stage. Individual proteins found in meconium can be a source of information pertaining to the intrauterine metabolic processes.Methods:
Concentrations of AAT, VDBP, calprotectin, myeloperoxidase, lactoferrin and elastase were measured using ELISA tests in 80 meconium samples collected from 19 healthy, full-term neonates.Results:
The meconium concentrations of VDBP and AAT (mean±SD, [mg/g meconium]: 3.74±6.93, 3.72±1.79, respectively) were approximately 1000 times higher than those of the protein granule proteins calprotectin, myeloperoxidase, elastase and lactoferrin (mean ± SD, [µg/g meconium]: 285.7±215.8, 1.83±1.73, 1.72±2.70, 45.58±78.89, respectively). The correlation between VDBP and AAT was negative (r= - 0.40. p=0.000) and those between VDBP and calprotectin (r=0.38, p=0.000) and VDBP and myeloperoxidase (r=0.45, p=0.000) were positive. AAT was found to correlate positively with lactoferrin (r=0.38, p=0.000).Conclusion:
The correlations between the concentrations of VDBP and AAT, and with neutrophil granule proteins observed in meconium indicate their functional relationship in the intrauterine environment of the developing fetus. Meconium can be seen as an apparently underutilized source of biomarkers for evaluation of metabolic processes specific to fetal development.Introduction
Neutrophils are one of the host of innate immune cells detected at the maternal-fetal interface. Careful regulation of the inflammatory and immunoregulatory functions of these cells most likely plays a role in successful gestation [1, 2]. Still, neutrophil assays are not commonly used in perinatal diagnosis as there are no specific biological materials available for their detection that could be simply and noninvasively obtained from the intrauterine environment. However, neutrophil granule proteins detected and measured in meconium seem a practical alternative that could help to better understand the biological role(s) of neutrophils and their involvement in normal and disease processes during fetal development.
Meconium, which is a clinical material specific to gestation and fetal development, begins to appear in the fetal bowel at 13 weeks of gestation and accumulates until birth. As the first feces of the newborn it is noninvasively collected in the first 24-48 hours after birth. It is a carrier of numerous chemical compounds and metabolites, including specific proteins with different biological properties and functions during gestation [ 3, 4]. In the current laboratory diagnostics, the potential properties of meconium as a source of specific parameters for assessing the importance of intrauterine development on the health of the newborn are not sufficiently understood or practically used.
Proteomics has confirmed the complexity of meconium proteins [3, 4]. Selection of certain individual proteins detected in meconium, which interact with each other in the performance of their biological functions, may offer a new diagnostic panel and provide knowledge about their role(s) in maintaining homeostasis of the intrauterine environment.
This paper presents the associations between the abundant extracellular proteins vitamin D-binding protein (VDBP) and alpha-1 antitrypsin (AAT) and the neutrophil cytosolic proteins calprotectin, myeloperoxidase, elastase and lactoferrin detected in the meconium of healthy, full-term neonates, supported by a review of the available experimental evidence of their individual properties and interactions with each other.
VDBP (58 kDa), an abundant serum protein linked to numerous biological processes during pregnancy, is a glycosylated α2 – globulin produced in the liver [ 5, 6, 7, 8, 9]. VDBP is bound to specific, activated regions on the neutrophil plasma membrane using myeloperoxidase released from the azurophilic granules [10]. VDBP shedding from neutrophils is mediated by intracellular elastase and inhibited by AAT, which leads to VDBP accumulation on the neutrophil plasma membrane [11, 12, 13].
AAT (52.0 kDa), an abundant serum protein, single-chain glycoprotein, α1 – globulin produced in the liver. It is a serine protease inhibitor, which inhibits a range of proteases derived from degranulated neutrophils including neutrophil elastase. AAT inhibits VDBP shedding from neutrophils [12, 14, 15].
Calprotectin (36.5 kDa), a heterodimeric (S100A8/A9) calcium- and zinc-binding protein which makes up 5 % of the total protein and 60% of the cytosolic protein of neutrophils. It is present in meconium at higher (even >10 fold) levels than in feces of children over 1 year of age and adults [3, 16].
Myeloperoxidase (150.0 kDa), an inflammatory enzyme stored in the azurophilic granules of the neutrophil, generates reactive hypochlorous acid and chloride ions and modifies biomolecules in hosts during excess inflammation with broad-range specificity. The highly specific binding of VDBP by MPO has been linked to the increased VDBP uptake by activated neutrophils [17].
Elastase (29.5 kDa), a protease stored in the azurophilic granules of the neutrophil, controls the amount of VDBP bound to the cell surface by shedding of intact forms of VDBP from the cell surface into the extracellular environment [12]. Inhibition of elastase activity by AAT results in increased accumulation of VDBP on the neutrophil surface [15].
Lactoferrin (76.0 kDa), an iron-binding glycoprotein found in the secondary granules of the polymorphonuclear granulocytes [4]. Significant positive correlation between plasma lactoferrin concentrations and the elastase: AAT complex was associated with an increase in degranulation of activated neutrophils [18, 19].
The aim of the study was to confirm the experimental interdependencies between the abundant extracellular proteins (VDBP and AAT) and the neutrophil granule proteins (calprotectin, lactoferrin, myeloperoxidase and elastase) in meconium of healthy, full-term neonates as a new opportunity to provide knowledge about processes during intrauterine development.
Materials and Methods
The study material consisted of 80 meconium samples collected from the nappy with a disposable spatula and transferred into 50-ml graduated plastic containers. The weight of the total amount of meconium [g] and the length of time it was excreted after birth differed between the newborns. The total amount of meconium was taken from each newborn approximately within the first 2 days after birth in two to four consecutive portions. The date, time and weight of each collection were recorded. Meconium collection was considered complete when on inspection a dark-greenish-black color of this material with characteristic gel-like consistency had changed to the yellow-brown color characteristic of stool. The collected meconium samples were frozen at - 20ºC for 3 - 5 days and subsequently stored at - 80ºC for up to 3 months.
Neonates
Nineteen (19) neonates born in the Clinical Department of Obstetrics, Female Diseases and Gynecological Oncology, Central Clinical Hospital of the Ministry of the Interior, Warsaw, were included in the study.
Neonatal profile: gestational age [weeks]: mean±SD: 38.6 ±1.3 (range 36-41); birth weight [g]: mean±SD: 3317.4±553.5 (range 2040-4280); Apgar Score [min 1/3/5/10]: 10/10/10/10 (n=15), 9/10/10/10 (n=1), 9/9/10/10 (n=1), 8/9/10/10 (n-1), 9/9/9/9 (n=1).
Methods
Assay of proteins.
Measurements of meconium protein concentrations were performed according to the manufacturer`s instructions, using following kits:
- for Vitamin D-binding protein: The Human (DBP) ELISA kit, SunRed (Shanghai, www.sunredbio.com)
- for AAT: (Alpha-1-Antitrypsin) DRG Instruments GmbH, Marburg, Germany
- for calprotectin: the PhiCal Calprotectin ELISA Kit (MRP 8/14), Immudiagnostik AG
- for myeloperoxidase: the Myeloperoxidase (stool, urine) ELISA kit, DRG Instruments GmbH, Marburg, Germany
- for elastase: the Human PMN-elastase ELISA, BioVendor Research and Diagnostic Products
- for lactoferrin: The Human Lactoferrin ELISA kit, Assaypro LLC, St. Charles, MO
Statistical analysis
Statistical analysis was performed using the STATISTICA software, version 13.0. Results are reported as mean, standard deviation (SD), coefficient of variation (CV), median and range.
Correlations between meconium VDBP, AAT, calprotectin, myeloperoxidase, elastase and lactoferrin were assessed with the Spearman test and statistically significant differences were assumed when p<0.05.
Results
Table 1 presents the concentrations of VDBP, AAT and neutrophil granule proteins (calprotectin, myeloperoxidase, elastase and lactoferrin) in meconium collected from healthy, full-term neonates.
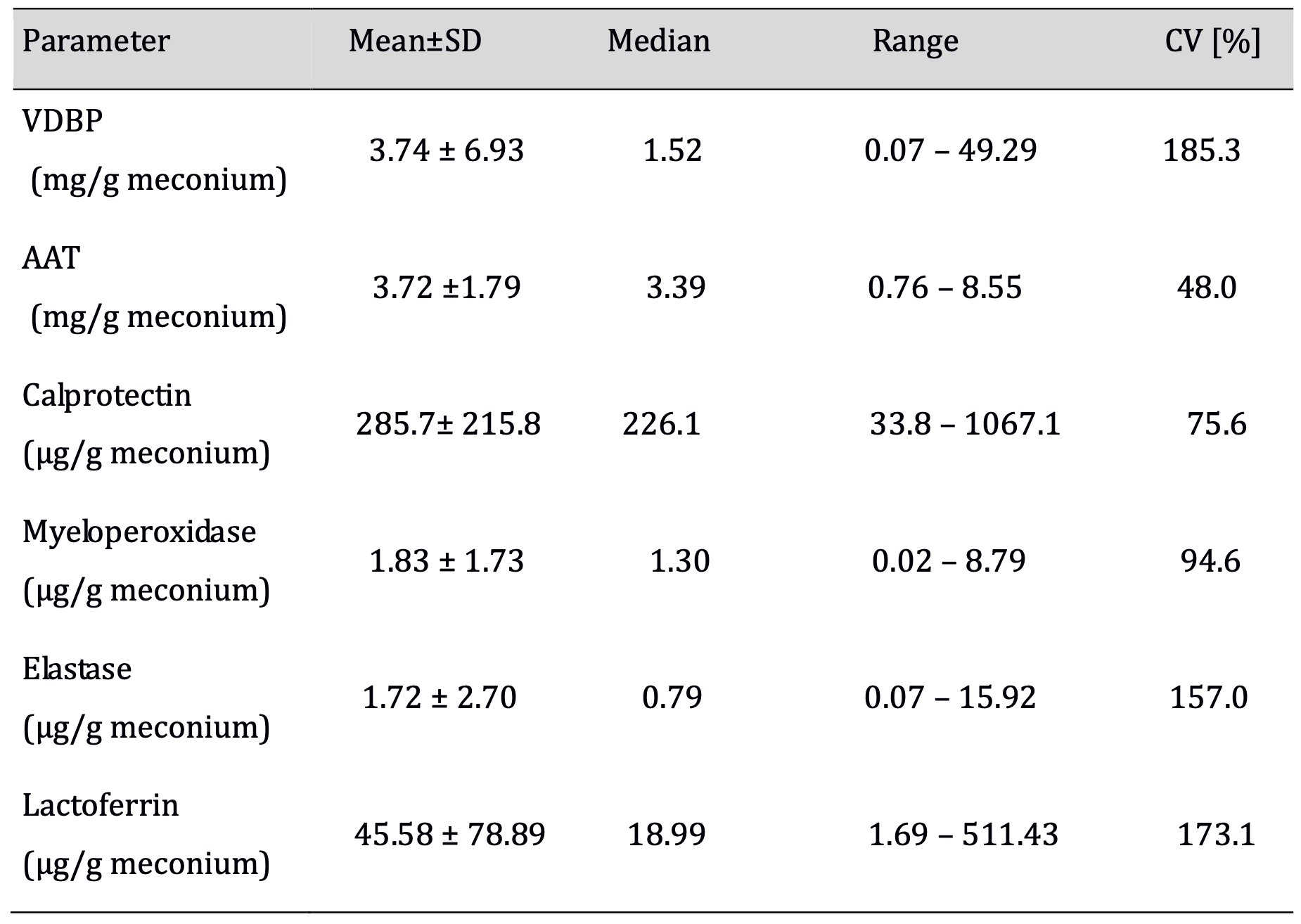
Table 1: Concentrations of VDBP, AAT, and neutrophil granule proteins in meconium samples collected from 80 healthy, full-term neonates. CV% = coefficient of variation
The meconium concentrations of VDBP and AAT were similar, with a higher dispersion of values and coefficient of variation for VDBP.
The meconium concentrations of neutrophil granule proteins were 1000 times lower than those of VDBP and AAT. The coefficients of variation for elastase and lactoferrin were more than twice as high as those for calprotectin and myeloperoxidase.
The correlations between VDBP, AAT and neutrophil granule proteins are presented in Table 2.
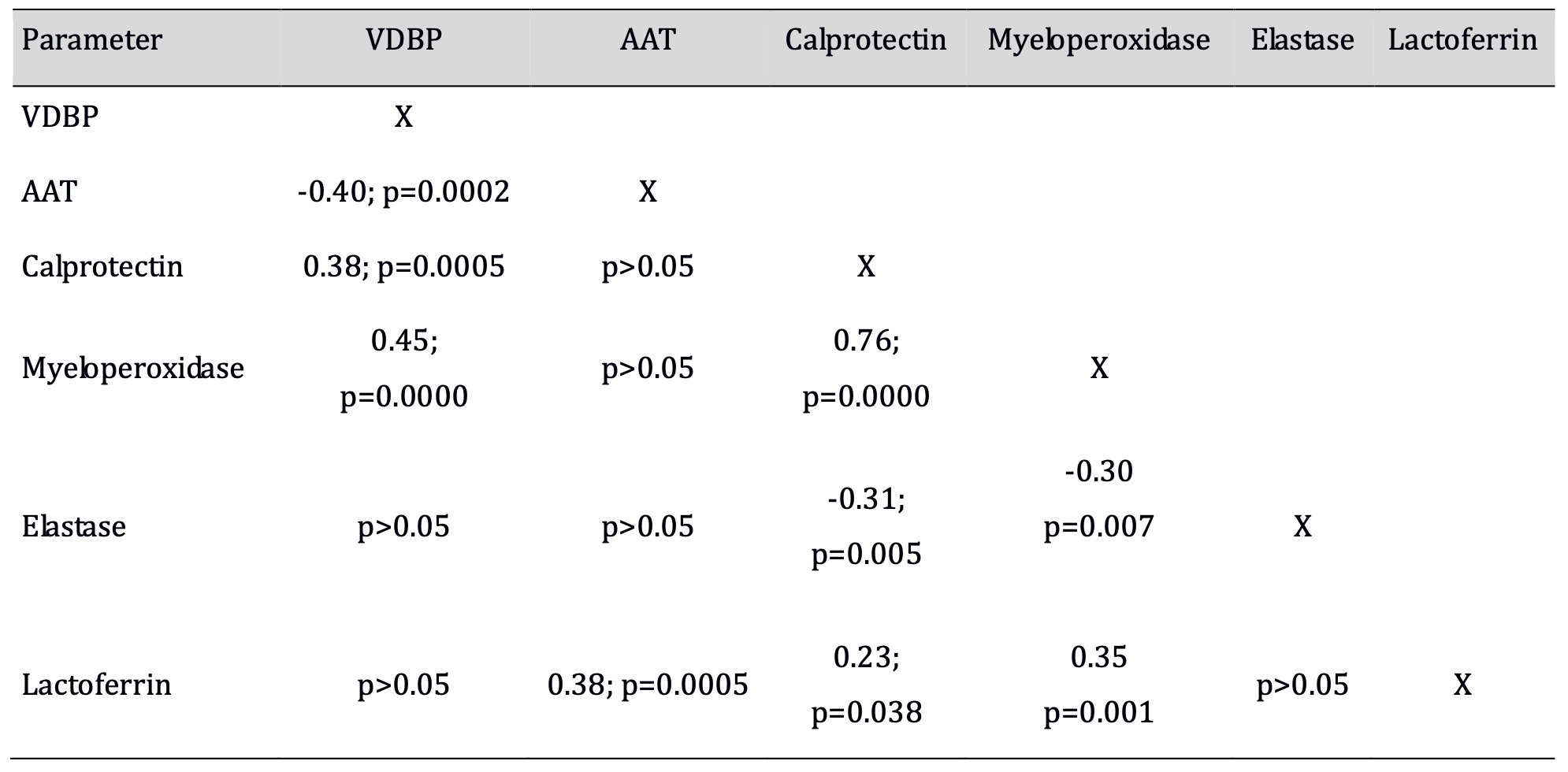
Table 2: Correlation coefficients between the concentrations of VDBP, AAT, and neutrophil granule proteins. Significant correlations (p<0.05) in bold type
A significant negative correlation (p<0.05) was established between the meconium concentrations of VDBP and AAT. The concentrations of VDBP and AAT correlated with individual neutrophil granule proteins. There were significant positive correlations (p<0.05) between the concentrations of VDBP and those of myeloperoxidase and calprotectin, and of AAT with lactoferrin. Significant correlations (p<0.05) were found between the concentrations of calprotectin, myeloperoxidase, elastase and lactoferrin.
The scattergraph (Fig. 1) shows the associations between the meconium concentrations of VDBP and those of AAT, calprotectin and myeloperoxidase established in 80 meconium samples from healthy, full-term neonates. There was a negative correlation between VDBP and AAT, and positive correlations between VDBP and myeloperoxidase and between VDBP and calprotectin.
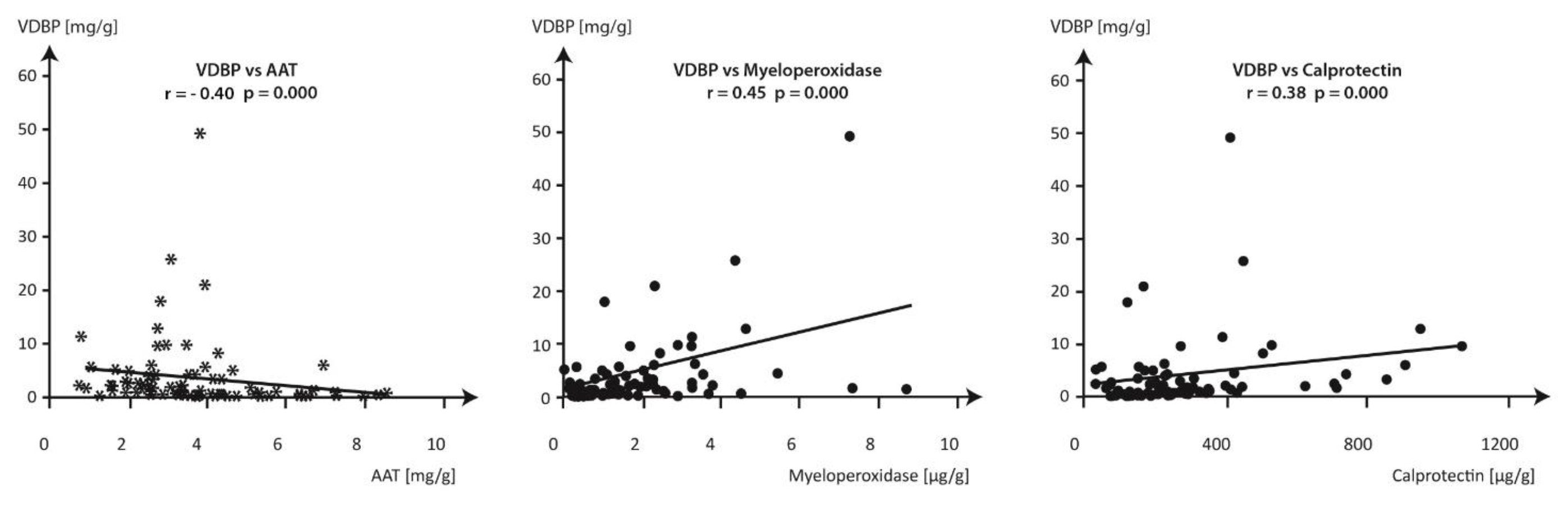
Fig. 1: Correlations between the concentrations of VDBP, AAT, calprotectin and myeloperoxidase in meconium samples collected from healthy, full-term infants. * negative correlation between VDBP and AAT r= - 0.40, p=0.000 ● positive correlations between VDBP and myeloperoxidase r=0.45, p=0.000 and between VDBP and calprotectin r=0.38 , p=0.000
The meconium VDBP concentrations varied over a wide range from 0.07 mg/g to 49.29 mg/g meconium. In 30 meconium samples (37.5%), the concentrations of VDBP were below 1 mg/g meconium with a concomitant high dispersion of the measurements of AAT (0.93-8.55 mg/g), myeloperoxidase (0.22 – 4.55 µg/g) and calprotectin (75.7-432.0 µg/g). The scattergraph confirms the associations between meconium proteins established when the concentration of VDBP exceeded 1 mg/g meconium.
Discussion
The findings suggest potential new approaches to the identification of complex normal and disease processes in utero using individual proteins measured in meconium.
Meconium passed after birth is an easily available biological material, which however is underutilized in laboratory diagnostics. Meconium begins to appear in the fetal bowel at 13 weeks of gestation and accumulates until birth and is excreted as the first feces of the newborn after birth. It carries numerous chemical compounds and metabolites, including individual proteins from swallowed amniotic fluid, shed intestinal epithelial cells and intestinal secretions. As a result, it is a reservoir of biomarkers derived from the intrauterine environment [3, 4]. In response to the clinical need, we wanted to identify the biomarkers that could combine the diagnostic information from the intrauterine environment in which the fetus develops with the clinical assessment of the neonate performed after birth. In view of the knowledge gained from earlier experimental studies by other authors [10, 12], the series of innate-immunity proteins assessed in this study which are involved in the regulation of neutrophils, may confirm their role(s) in meconium in vivo, as an etiologic factor present in the intrauterine environment, but possibly implicated in later disease. According to the fetal origins of adult disease (FOAD) hypothesis [20] it may improve our understanding of the effects of intrauterine environmental exposures during sensitive periods in fetal development with a possibly increased risk of specific diseases in adult life.
It is generally agreed [1, 2, 13] that neutrophils play a fundamental role in successful pregnancy outcome, but their exact function in the intrauterine microenvironment remains unclear. Both fetal and maternal neutrophils can invade the amniotic cavity, suggesting that both the fetus and the mother participate in the cross-talk between neutrophils and microenvironment at the intrauterine level [1, 2]. Neutrophils are the primary mediators of the rapid immune response. However, relatively little is known about the impact of physiological and pathological components of body fluids on their recruitment into tissues and targeted neutrophil response [10, 11, 12].
A negative correlation between the meconium concentrations of AAT and VDBP may reasonably suggest mutual regulation existing between these abundant extracellular proteins to maintain homeostasis of the intrauterine environment. The findings of this study according to experimental observations of other authors [10, 11, 12] may help to elucidate the biological role of VDBP and AAT in the intrauterine environment for regulating the level of neutrophil granules proteins. The characteristic correlation between the levels of VDBP and myeloperoxidase in the meconium confirms in vivo the experimental findings of other authors [11, 12, 17] identifying myeloperoxidase found in the azurophilic granules of neutrophils as a reservoir of VDBP binding sites on the surface of neutrophils and suggests similar processes originating in utero.
A positive correlation between VDBP and calprotectin may on the one hand suggest a common biological role shared by the extracellular and the intraneutrophil proteins in the regulation of local inflammation. On the other hand, considering the abundance of calprotectin in neutrophils, the assessment of calprotectin concentration and its correlations with other proteins in meconium, can be a practical alternative to the measurement of neutrophil count and the proportion of neutrophils binding VDBP in the intrauterine environment [4, 16].
Azurophilic granules of the neutrophil are the activated binding site for VDBP using myeloperoxidase, but also the location of intracellular elastase. Published studies [12] emphasize the key role of elastase in the mechanism of VDBP shedding from neutrophils into the extracellular environment, but we found no correlation between the concentrations of VDBP and elastase in meconium. The sequence of maintaining equilibrium between the quantity of neutrophil-bound VDBP and elastase activity indirectly involves AAT as a potential elastase inhibitor whose concentration in meconium is more than 1000 times higher than that of elastase concentration [10, 12, 15]. A positive correlation of AAT concentrations with those of lactoferrin may suggest selective mechanisms of neutrophil degranulation as a result of their activation.
Given that VDBP binds to the activated sites on neutrophils [10, 11], positive correlations with myeloperoxidase and calprotectin and negative correlations with AAT become significant with the increasing VDBP concentrations. The graphical presentation of the relationship between VDBP and other proteins in Fig. 1 confirms their deepening at a concentration of VDBP in meconium above 1 mg/g meconium. The obtained results indicate a clear need to determine a rigorous range of VDBP concentration in meconium, which may increase the relationship between these proteins. The small number of newborns presented in this paper prevents statistical support for reported findings.
According to the authors [5, 21, 22, 23, 24], the characteristic properties of the proteins presented in this work are the basis for their potential use for early assessment of the health of newborns and adults. Genetically determined AAT deficiency can cause chronic liver diseases, cirrhosis, and liver failure in children and adults, and lung diseases in adults but it is underrecognized and most patients are undiagnosed [21]. Low concentrations of maternal serum VDBP concentrations have been associated with potential effects on the pathogenesis of type 1 diabetes mellitus in the offspring, preeclampsia, preterm birth and fetal growth restriction [5]. Assessment of myeloperoxidase [22] and elastase alpha1 proteinase inhibitor complex [23] enables detection of neonatal sepsis, supplementation lactoferrin [24] prevent sepsis and necrotizing enterocolitis in preterm infants. Differences in the mechanisms underlying the effects of VDBP and AAT on the biological function of neutrophil granule proteins in meconium raises the question concerning the diagnostic potential of these observations in differentiating between normal and disease conditions in utero. As observed by other authors [2, 8], VDBP binding to the activated sites on neutrophils is an important stage in their role in the immune system as chemotactic activity of complement (C) peptides C5a and C5a des arg is enhanced [10, 13]. AAT has a key role in the innate immune response, possesses an anti-inflammatory capacity, inhibits VDBP shedding from neutrophils and its serving as a chemotactic co- factor for C5a [12, 14, 15]. Calprotectin released from activated neutrophils increases its concentration mainly in inflammatory processes and may be a sensitive indicator of local inflammation [3, 16]. Lactoferrin plays a key role in the innate immunity response and has antimicrobial effects [4]. This observation may account for a tight regulation of neutrophil proinflammatory functions in the intrauterine environment with high VDBP and AAT concentrations in the extracellular space [11]. Further studies of the association of these proteins with markers of innate immune cells in meconium and maternal peripheral blood may provide insight into their potential diagnostic role.
Conclusion
In conclusion, the current knowledge of the biological roles individual proteins play in fetal development is limited and further studies are required. Meconium which is obtained non-invasively may be used for the post-natal laboratory evaluation of the fetal stage as it is a reservoir of intrauterine proteins. The selection of meconium proteins in this study was proposed as a potential diagnostic tool to elucidate the role of neutrophils in normal and disease processes in utero and to differentiate between these two aspects of their activity. The correlations between AAT and VDBP, and neutrophil granule proteins suggest that this protein panel could be used to retrospectively evaluate fetal development and diagnose and resolve health problems related the adverse intrauterine environments.Acknowledgements
We acknowledge Jennifer Doering and Ying-hui Rong of the Wadsworth Center (NYS Department of Health) for their technical insight.
Author Contributions
Barbara Lisowska-Myjak : the main conceptual ideas, supervised the findings of this work, wrote the manuscript; Ewa Skarżyńska : material preparation, data collection and analysis; Artur Jakimiuk : contributed to the design and implementation of the research. All authors read and approved the final manuscript., Artur Jakimiuk has received research support from Institute of Mother and Child.
Funding Sources
Grant from Institute of Mother and Child nr OPK 510-15-31
Ethics approvals
This study was performed in line with principles of the Declaration of Helsinki. Approval was granted by the Local Committee for Human Experiments at the Central Clinical Hospital of the Ministry of the Interior (Decision No. 71/2011).
Consent to participate
Informed consent was obtained from all individual participants included in the study. Informed consent was obtained from a parent.
Disclosure Statement
The authors have no conflicts of interest to declare.
References
| 1 | Gomez-Lopez N, Romero R, Xu Y, Leng Y, Garcia-Flores V, Miller D, Jacques SM, Hassan SS, Faro J, Alsamsam A, Alhousseini A, Gomez-Roberts H, Panaitescu B, Yeo l, Mayom E: Are amniotic fluid neutrophils in women with intraamniotic infection and/or inflammation of fetal or maternal origin? Am J Obstet Gyn 2017;6:693.e1-693.e16. https://doi.org/10.1016/j.ajog.2017.09.013 |
| 2 | Bert S, Ward EJ, Nadkarni S: Neutrophils in pregnancy: New insights into innate and adaptive immune regulation. Immunology 2021;164:665-676. https://doi.org/10.1111/imm.13392 |
| 3 | Lisowska-Myjak B, Żytyńska-Daniluk J, Skarżyńska E: Concentrations of neutrophil-derived proteins in meconium and their correlations. Biomark Med 2016;10:819-829. https://doi.org/10.2217/bmm-2016-0034 |
| 4 | Lisowska-Myjak B, Skarżyńska E, Wojdan K, Nasierowska-Guttmejer A: Protein and peptide profiles in neonatal meconium. J Obstet Gynaecol Res 2019;45:556-564. https://doi.org/10.1111/jog.13888 |
| 5 | Fernando M, Ellery SJ, Marquina C, Lim S, Naderpoor N, Mousa A: Vitamin D-binding protein in pregnancy and reproductive health. Nutrients 2020;12:1489. https://doi.org/10.3390/nu12051489 |
| 6 | Liong S, Di Quinzio MKW, Fleming G, Permezel M, Georgiou HM: Is vitamin D binding protein a novel predictor of labour? PLoS ONE 2013;8:e76490. https://doi.org/10.1371/journal.pone.0076490 |
| 7 | Fernando M, Ellery SJ, de Guingand D, Marquina C, Lim S, Harrison CL, Teede HJ, Naderpoor N, Mousa A: Early pregnancy vitamin D binding protein is independently associated with the development of gestational diabetes: a retrospective cohort study. J Clin Med 2020;9:2186. https://doi.org/10.3390/jcm9072186 |
| 8 | Ma R, Gu Y, Zhao S, Sun J, Groome LJ, Wang Y: Expressions of vitamin D metabolic components VDBP, CYP2R1, CYP27B1, CYP24A1, and VDR in placentas from normal and preeclamptic pregnancies. Am J Physiol Endocrinol Metab 2012;303:E928-E935. https://doi.org/10.1152/ajpendo.00279.2012 |
| 9 | Kook SY, Park KH, Jang JA, Kim YM, Park H, Jeon SJ: Vitamin D-binding protein in cervicovaginal fluid as a non-invasive predictor of intra amniotic infection and impending preterm delivery in women with preterm labor or preterm premature rupture of membranes. PLoS ONE 2018;13:e0198842. https://doi.org/10.1371/journal.pone.0198842 |
| 10 | Kew RR: The vitamin D binding protein and inflammatory injury: a mediator or sentinel of tissue damage? Front Endocrinol 2019;10:470. https://doi.org/10.3389/fendo.2019.00470 |
| 11 | DiMartino SJ, Trujillo G, McVoy LA, Zhang J, Kew RR: Upregulation of vitamin D binding protein (Gc-globulin) binding sites during neutrophil activation from latent reservoir in azurophil granules. Mol Immunol 2007;44:2370-2377. https://doi.org/10.1016/j.molimm.2006.10.011 |
| 12 | DiMartino SJ, Shah AB, Trujillo G, Kew RR: Elastase controls the binding of the vitamin D-binding protein (Gc-Globulin) to neutrophils: a potential role in the regulation of C5a co-chemotactic activity. J Immunol 2001;166:2688-2694. https://doi.org/10.4049/jimmunol.166.4.2688 |
| 13 | Karras SN, Koufakis T, Fakhoury H, Kotsa K: Deconvoluting the biological roles of vitamin D-binding protein during pregnancy: a both clinical and theoretical challenge. Front Endocrinol 2018;9:259. https://doi.org/10.3389/fendo.2018.00259 |
| 14 | Fee LT, Gogoi D, O'Brien ME, McHugh E, Casey M, Gough C, Murphy M, Hopkins AM, Carroll TP, McElvaney NG, Reeves EP: C3d elicits neutrophil degranulation and decreases endothelial cell migration, with implications for patients with alpha-1 antitrypsin deficiency. Biomedicines 2021;9:1925. https://doi.org/10.3390/biomedicines9121925 |
| 15 | Lindley VM, Bhusal K, Huning L, Levine SN, Jain SK: Reduced 25(OH) vitamin D association with lower alpha-1-antitrypsin blood levels in type 2 diabetic patients. J Am College of Nutrition 2021;40:98-103. https://doi.org/10.1080/07315724.2020.1740629 |
| 16 | Jerkic SP, Michel F, Donath H, Herrmann E, Schubert R, Rosewich M, Zielen S: Calprotectin as a new sensitive marker of neutrophilic inflammation in patients with bronchiolitis obliterans. Mediators Inflamm 2020; 4641585. https://doi.org/10.1155/2020/4641585 |
| 17 | Kato Y: Neutrophil myeloperoxidase and its substrates: formation of specific markers and reactive compounds during inflammation. J Clin Biochem Nutr 2016;58:99-104. https://doi.org/10.3164/jcbn.15-104 |
| 18 | Nuijens JH, Abbink JJ, Wachtfogel YT, Colman RW, Eerenberg AJ, Dors D, Kamp AJ, van Schijndel RJS, Thijs LG, Hack CE: Plasma elastase alpha 1-antitrypsin and lactoferrin in sepsis: evidence for neutrophils as mediators in fatal sepsis. J Lab Clin PutrMed 1992;119:159-168. |
| 19 | Janciauskiene S, Wrenger S, Immenschuh S, Olejnicka B, Greulich T, Welte T,Chorostowska-Wynimko J.: The Multifaceted Effects of Alpha1-Antitrypsin on Neutrophil Functions. Front. Pharmacol. 2018;9:341. https://doi.org/10.3389/fphar.2018.00341 |
| 20 | Skogen JC, Overland S: The fetal origins of adult disease: a narrative review of the epidemiological literature. JRSM Short Rep 2012;3:59. https://doi.org/10.1258/shorts.2012.012048 |
| 21 | Teckman J, Pardee E, Howell RR, Mannino D, Sharp RR, Brantly M, Wanner A, Lamson J. Alpha-1 Foundation Workshop Attendees: Appropriateness of newborn screening for α1-antitrypsin deficiency. J Pediatr Gastroenterol Nutr 2014;58:199-203. https://doi.org/10.1097/MPG.0000000000000196 |
| 22 | Yunanto A, Edi Hartoyo E, Thalib I, Putri NA, Suhartono E: Kinetic parameter analysis of myeloperoxidase in the presence of some cosusbtrates in saliva of newborn at risk of sepsis. Int J Applied Pharm 2021;13. https://doi.org/10.22159/ijap.2021.v13s1.Y0072 |
| 23 | Tegtmeyer FK, Horn C, Richter A, van Wees J: Elastase alpha 1 proteinase inhibitor complex, granulocyte count, ratio of immature to total granulocyte count, and C-reactive protein in neonatal septicaemia. Eur J Pediatr 1992;151:353-356. https://doi.org/10.1007/BF02113257 |
| 24 | Manzoni P, Dall'Agnola A, Tomé D, Kaufman DA, Tavella E, Pieretto M, Messina A, De Luca D, Bellaiche M, Mosca A, Piloquet H, Simeoni U, Picaud JC, Del Vecchio A: Role of Lactoferrin in Neonates and Infants: An Update. Am J Perinatol 2018;35:561-565. https://doi.org/10.1055/s-0038-1639359 |