Original Article – DOI: 10.33594/000000863
CPB (60): 213 - 222
Accepted: 08.04.2026 - Published: 27.04.2026
Acetylcholinesterase Activity in Erythrocytes: a Functional Sensor of Environmental Toxicity from Brominated Flame Retardants
bDepartment of Cytobiochemistry, Faculty of Biology and Environmental Protection, University of Lodz, Pomorska 141/143, 90-236 Lodz, Poland;
Keywords
Abstract
Background/Aims: Brominated flame retardants (BFRs) are a group of synthetic compounds widely used to reduce the flammability of various materials. Due to their large scale production and extensive industrial application, increasing concern has arisen regarding their persistence in the environment and potential toxicological effects. In an effort to identify novel biomarkers indicative of cellular dysfunction caused by BFRs exposure, the aim of the present study is to investigate the activity of acetylcholinesterase (AChE) in human erythrocytes exposed to selected compounds, including tetrabromobisphenol A (TBBPA), tetrabromobisphenol S (TBBPS), 2,4-dibromophenol (2,4-DBP), 2,4,6-tribromophenol (2,4,6-TBP), and pentabromophenol (PBP). Methods: AChE activity was determined spectrophotometrically using Ellman’s method after incubation of erythrocytes with the tested compounds. Results: The obtained results demonstrated that both TBBPA and TBBPS induced significant alterations in AChE activity. A statistically significant increase in enzyme activity was observed at concentrations of 10 µg/mL for TBBPA and 50 µg/mL for TBBPS. In the case of bromophenols, significant changes were detected only for 2,4,6-TBP at 25 µg/mL and PBP at 10 µg/mL. Conclusion: The results indicate that the biological effects of BFRs depend on their chemical structure and concentration. Although erythrocyte AChE activity responds to BFR exposure, it should not be regarded as a highly sensitive biosensor, since detectable alterations occur at concentrations that simultaneously elicit other cellular effects, including lipid peroxidation, phosphatidylserine externalization, and modifications in membrane fluidity, as reported in previous studies.
Introduction
Brominated flame retardants (BFRs) is a group of synthetic chemical substances used to reduce the flammability of organic materials and the risk of ignition and flame spread. The need to enhancement of fire hazard prevention has resulted in the widespread application of these compounds across various consumer products such as textiles, electrical and electronic equipment, wood preservatives, plastics and other [1]. The discussed group of compounds includes over 80 different chemical substances. Many of these compounds (e.g., certain PBDEs and HBCD), due to their propensity for bioaccumulation and their potential impact on human health and the environment, have been subjected to legal regulations and, in some cases, withdrawn from use or replaced with less hazardous alternatives. At present, tetrabromobisphenol A (TBBPA) is the most widely applied compound within this group, accounting for approximately 60% of the brominated flame retardant (BFR) market and about 25% of the global flame-retardant market [2]. According to REACH registration data, TBBPA is produced or imported into the European Economic Area in quantities of 10, 000–100, 000 tonnes per year. Considering this scale of production and its extensive use in industrial applications, increasing attention has been directed towards its environmental persistence and toxicological effects. In 2023, TBBPA was added to the Candidate List of substances of very high concern (SVHC) due to its classification as a Category 1B carcinogen [3, 4]. There is growing evidence that TBBPA exert toxic effects on multiple biological systems. Experimental studies have shown their potential to disrupt endocrine signalling, impair neurodevelopment, induce oxidative stress, and modulate immune responses [5-10]. As a consequence, novel analogues of TBBPA such as tetrabromobisphenol S (TBBPS) are being introduced into industrial applications. Despite the widespread occurrence of TBBPA and its derivatives in the environment as well as in human and animal organisms, and their potential toxic effects, no regulations limiting the production of TBBPA or its derivatives have been implemented to date, either within the European Union or globally [11]. Erythrocytes, although lacking a nucleus, play a pivotal role in oxygen transport and in maintaining microcirculatory homeostasis, which makes them particularly susceptible to alterations induced by toxic compounds. Acetylcholinesterase (AChE), best known for its role in neurotransmission, is also abundantly present in erythrocytes. The activity of membrane-bound AChE in red blood cells serves as a sensitive indicator of cell membrane integrity and functionality, and its perturbation can reflect early stages of cellular injury, including oxidative stress and the initiation of eryptosis [12, 13]. Our previous studies demonstrated that the studied BFRs modulate signalling pathways leading to eryptosis, as evidenced by, among other findings, increased intracellular calcium levels (PBP, 50 µg/mL), enhanced phosphatidylserine externalization (TBBPA, 2, 4-DBP, 2, 4,6-TBP, PBP from 10 µg/mL, TBBPS from 100 µg/mL), caspase-3 activation (most prominently for TBBPA from 1 µg/mL), and reduced calpain levels (for TBBPA at 100 µg/mL and PBP from 50 µg/mL) [13]. Moreover, these compounds were found to compromise the antioxidant defence system by decreasing the activities of SOD, CAT, and GSH-Px to varying degrees depending on the specific BFR and applied concentration, and they induced ROS generation even at very low concentrations [12, 13]. Seeking additional markers capable of indicating cellular dysfunction elicited by these BFRs, the present study aims to evaluate AChE activity in erythrocytes exposed to selected compounds, such as tetrabromobisphenol A (TBBPA), tetrabromobisphenol S (TBBPS), 2, 4-dibromophenol, 2, 4,6-tribromophenol, and pentabromophenol (Fig. 1). A broad concentration range (1–250 µg/mL) was applied, within which no hemolysis or only trace hemolysis had been previously observed upon exposure to brominated compounds [14]. The maximum incubation time of 48 h was chosen based on earlier experiments with erythrocytes, reflecting the established stability of these anucleate cells during storage.
Fig. 1: The chemical formulas of studied BFRs. Created in BioRender. Jarosiewicz, M. (2025) https://BioRender.com/ln3qy7x.
Materials and Methods
Chemicals
TBBPA (purity 99%, 2, 6-dibromo-4-[2-(3, 5-dibromo-4-hydroxyphenyl)propan-2-yl]phenol)) was purchased from LGC Standards (Germany). Tetrabromobisphenol S (purity 98.8%, 2, 6-dibromo-4-(3, 5-dibromo-4-hydroxyphenyl)sulfonylphenol) was synthetized in the Institute of Industrial Organic Chemistry in Warsaw in Poland. 2, 4-DBP (98% purity) and 2, 3,4, 5,6-PBP (98% purity), were obtained from LGC Standards (Germany). 2, 4,6-TBP (purity ≤ 100%) and acetylthiocholine iodide were purchased from Sigma-Aldrich (USA). Other chemicals were bought from Roth (Germany) or POCh (Poland).
Erythrocyte isolation and treatment
Human red blood cells (RBCs) were obtained from the leukocyte- buffy coat, separated from blood collected from healthy volunteers at the Regional Centre for Blood Donation and Blood Treatment in Lodz, Poland. The method for RBCs isolation and preparation followed the protocol previously described in Jarosiewicz et al. (2017). RBCs were adjusted to a hematocrit of 5% (approximately 630 million cells/mL) and incubated at 37 °C for 48 hours with the tested compounds. The applied concentration ranges were 1–25 µg/mL for TBBPA, 1–250 µg/mL for TBBPS, and 1–50 µg/mL for bromophenols. All compounds were dissolved in DMSO, with a final concentration of 0.4%. The concentrations were selected based on previously reported hemolytic activity [14]. For the 48-hour incubation, antibiotics (0.2% streptomycin and penicillin) were included to prevent microbial contamination. Control samples were prepared to verify that neither DMSO nor the antibiotics affected RBC integrity.
Acetylcholinesterase activity
The activity of acetylcholinesterase (AChE) was determined using a modified version of the method described by Ellman et al. (1961) [15], which relies on the enzymatic hydrolysis of acetylthiocholine iodide, resulting in the formation of acetic acid and thiocholine. Erythrocyte samples were diluted in 5 mM phosphate buffer (pH 8.0) to achieve a final hematocrit of 0.05%. To 2 mL of the diluted cell suspension, 20 μL of 10 mM DTNB prepared in 18 mM NaHCO₃ was added. Just before starting the measurement, 60 μL of acetylthiocholine iodide (10 mM) was introduced into the cuvette containing the reaction mixture. The change in absorbance was monitored for 1 minute at 412 nm using a Specord 250 Plus spectrophotometer (Analytik Jena AG). One unit of acetylcholinesterase (AChE) activity was defined as the number of micromoles of acetylthiocholine hydrolyzed per minute by the enzyme present in 1 mL of packed erythrocytes (100% hematocrit). Enzyme activity in experimental samples was expressed as a percentage of the control value. In the current study, the mean AChE activity in the control sample after 48 hours of incubation was 5.17 µmol/min/mL of packed cells.
Statistical analysis
Statistical analysis was performed using a parametric approach. The normality of data distribution was assessed prior to further analysis. One-way analysis of variance (ANOVA) was conducted, followed by Tukey's post hoc test to determine significant differences between groups. Results were considered statistically significant at p < 0.05. Data are presented as mean ± standard deviation (SD) from 4–6 individual experiments, each representing erythrocytes from a different donor. Additionally, erythrocytes from each donor were measured in six replicates. All statistical analyses were performed using Statistica and GraphPad Prism software.
Results
It was found that both TBBPA and TBBPS induced alterations in AChE activity in human erythrocytes. A statistically significant increase in AChE activity under the influence of TBBPA was observed starting from a concentration of 10 µg/mL, whereas TBBPS caused an increase in this parameter beginning at 50 µg/mL. The lack of changes in AChE activity at the concentration of 25 µg/mL may have resulted from hemolysis occurring at this concentration [14]. In the case of bromophenols, a statistically significant increase in AChE activity was observed only at a single concentration 25 µg/mL for 2, 4,6-TBP and 10 µg/mL for PBP. No concentration-dependent relationship was found in the enzyme activity changes induced by bromophenols (Fig. 2).
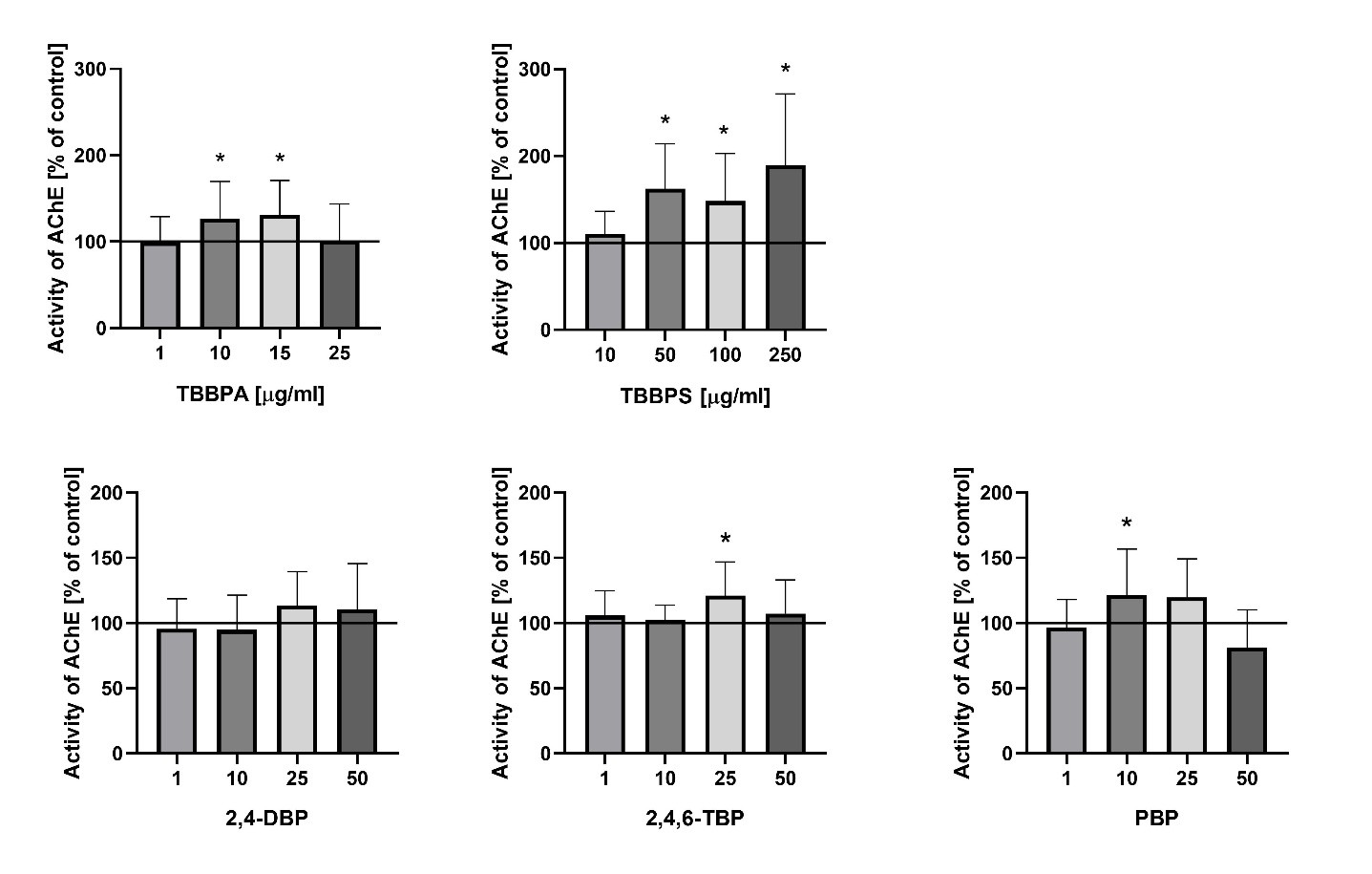
Fig. 2: Acetylcholinesterase (AChE) activity in human erythrocytes after 48 h exposure to selected BFRs (TBBPA, TBBPS, 2,4,6-TBP, PBP) at concentrations ranging from 1 to 250 µg/mL. Values are presented as mean ± SD. * Statistically significant differences compared to the control (p<0.05).
Discussion
Acetylcholinesterase (AChE) is an enzyme responsible for the hydrolysis of acetylcholine into choline and acetic acid, playing a crucial role in terminating cholinergic neurotransmission. Although its activity is particularly important in the nervous system, the form of AChE present in human erythrocytes represents a well-characterized and easily accessible model for evaluating the impact of various environmental and toxic factors on enzymatic function [16, 17]. This is due to the fact that erythrocytes participate in multiple signaling pathways, including eryptosis and pathways associated with oxidative stress. Moreover, erythrocytic AChE serves as a biomarker of cell membrane integrity [18]. Alterations in the activity of this enzyme have also been observed in the course of numerous diseases, including leukemia, phenylketonuria, inflammatory processes, neurological disorders, hyperlipidemia, and osteoporosis [19]. The form of AChE present in erythrocytes, anchored in the cell membrane, exhibits structural similarity to the neuronal form of the enzyme, although its biological function is not directly related to neurotransmission. In the present study, the effect of the tested brominated phenolic compounds on AChE activity in erythrocytes was found to be variable and dependent on both the type of compound and its concentration. The most pronounced effect was observed for TBBPA and TBBPS, which significantly increased enzymatic activity compared with the control. This effect may be associated with oxidative stress induced by these compounds and a potential increase in the exposure of this phosphatidylinositol-anchored enzyme on the outer cell surface due to changes in membrane fluidity [20]. A similar, although less pronounced, effect was also observed for 2, 4,6-TBP and PBP, where an increase in enzyme activity occurred only at specific concentrations. An increase in erythrocyte AChE activity has also been reported in other studies investigating bisphenol A and bisphenol S structurally similar compounds that do not contain bromine atoms in their structure. These researchers demonstrated an increase in AChE activity in human erythrocytes following 24 h incubation with bisphenols, with significant changes observed starting at a concentration of 5 µg/mL for both bisphenol A (BPA) and bisphenol S (BPS) [21]. An increase in AChE activity has also been demonstrated in animal models. In mussels, exposure to high concentrations of TBBPA resulted in elevated activity of both antioxidant enzymes (including SOD and CAT) and AChE [22]. Similarly, Wang and Dai (2022) showed that oral administration of TBBPA to mice for 28 days at doses of 50 or 100 mg/kg body weight led to a significant increase in AChE levels at the higher dose compared with the control group [23]. However, in in vivo systems, the mechanism of enzyme activation may differ substantially, potentially involving increased gene expression rather than changes in erythrocyte membrane properties and redox balance, as observed in in vitro conditions. Publicly available transcriptomic data from bisphenol A-exposed human Ishikawa cells (GEO dataset GDS4926; Naciff et al., 2010) indicate broad dose and time dependent gene expression changes, whereas ACHE expression itself does not show a consistent pattern of regulation. Although obtained in a different cellular model, these findings may support the hypothesis that the increased AChE activity observed in erythrocytes is more likely associated with post-transcriptional, membrane-related, and oxidative mechanisms rather than with direct transcriptional upregulation of ACHE [24]. Therefore, these data should be considered as supportive context rather than direct validation of the mechanisms observed in erythrocytes as they originate from a different cellular system with distinct exposure conditions. Our previous studies confirmed that the analyzed compounds modify the physical properties of the erythrocyte membrane by disrupting the fluidity of the hydrophilic region and inducing conformational changes in membrane proteins [25, 20, 25] (Table 1). Such modifications may alter the enzymatic microenvironment of AChE, leading to its increased exposure on the cell surface and subsequent activation. An increase in lipid peroxidation was also observed, indicating enhanced oxidative stress [20, 25]. AChE, being strongly associated with the cell membrane, is particularly sensitive to such alterations, and its elevated activity may be a consequence of these oxidative changes (Table 1). Another important aspect is the potential interaction with band 3 protein, which plays a crucial role in maintaining the structure of the erythrocyte cytoskeleton. The literature indicates that increased AChE activity in erythrocytes correlates with band 3 phosphorylation and enhanced cell aggregation [27]. Since the studied BFRs can modulate cytoskeletal structure through alterations in membrane fluidity and erythrocyte morphology [20, 26], it is likely that the observed changes in AChE activity may be indirectly regulated by the interaction of these compounds with the membrane protein–lipid system. Moreover, previous studies have shown that changes in glutathione (GSH) levels and other oxidative stress markers may modulate AChE activity by affecting membrane integrity through mechanisms involving nitric oxide (NO) signaling and the cellular redox state [18]. In addition to the role of oxidative stress and modifications of erythrocyte membrane physical properties, increased AChE activity may also be associated with other cellular processes. One possible mechanism is the induction of eryptosis, the programmed death of erythrocytes. It has been demonstrated that certain environmental toxins can trigger eryptosis, one of whose markers is increased AChE activity, along with the appearance of extracellular vesicles in plasma enriched with AChE derived from the membranes of senescent erythrocytes [28, 29]. This may suggest that the observed increase in AChE activity following exposure to BFRs could be associated with processes related to eryptosis, serving as a signal for the removal of affected erythrocytes from circulation. Previous results support this hypothesis, indicating that the studied BFRs may induce eryptosis through mechanisms involving disturbances in calcium homeostasis, phosphatidylserine exposure, caspase-3 activation, and alterations in calpain activity. Erythrocyte membrane-bound AChE is considered as an indicator of alterations in membrane integrity and overall homeostatic status, and its activity may reflect early effects of xenobiotic exposure. The observed changes suggest that AChE activity could serve as a potential biomarker of exposure to BFRs, particularly in environmental contexts where chronic exposure to low doses is likely. The increase in AChE activity, in contrast to the classical inhibition observed with organophosphate pesticides, points to a distinct mechanism of BFR toxicity that warrants further investigation in both cellular and in vivo models. In summary, the increase in AChE activity under exposure to the studied BFRs may be associated with several processes, including: destabilization of membrane structure (altered fluidity and protein conformation), induction of oxidative stress (lipid peroxidation and ROS generation), and activation of adaptive enzymatic responses aimed at maintaining cellular integrity (Fig. 3). Thus, the pattern of AChE modulation presented here, combined with the documented susceptibility of erythrocyte membranes to BFR-induced stress, suggests that AChE may have complementary diagnostic value in the environmental assessment of exposure to brominated contaminants, particularly when interpreted alongside additional markers of oxidative and eryptotic dysregulation [12, 13, 20, 26]. Nevertheless, it should be noted that the present study is based on a relatively limited experimental dataset and focuses on a single in vitro model of human erythrocytes. Therefore, the findings should be interpreted as a targeted investigation providing initial insights into the effects of BFRs on AChE activity, rather than a comprehensive mechanistic analysis. Further studies, particularly in more complex cellular systems and in vivo models, are required to confirm the proposed mechanisms.
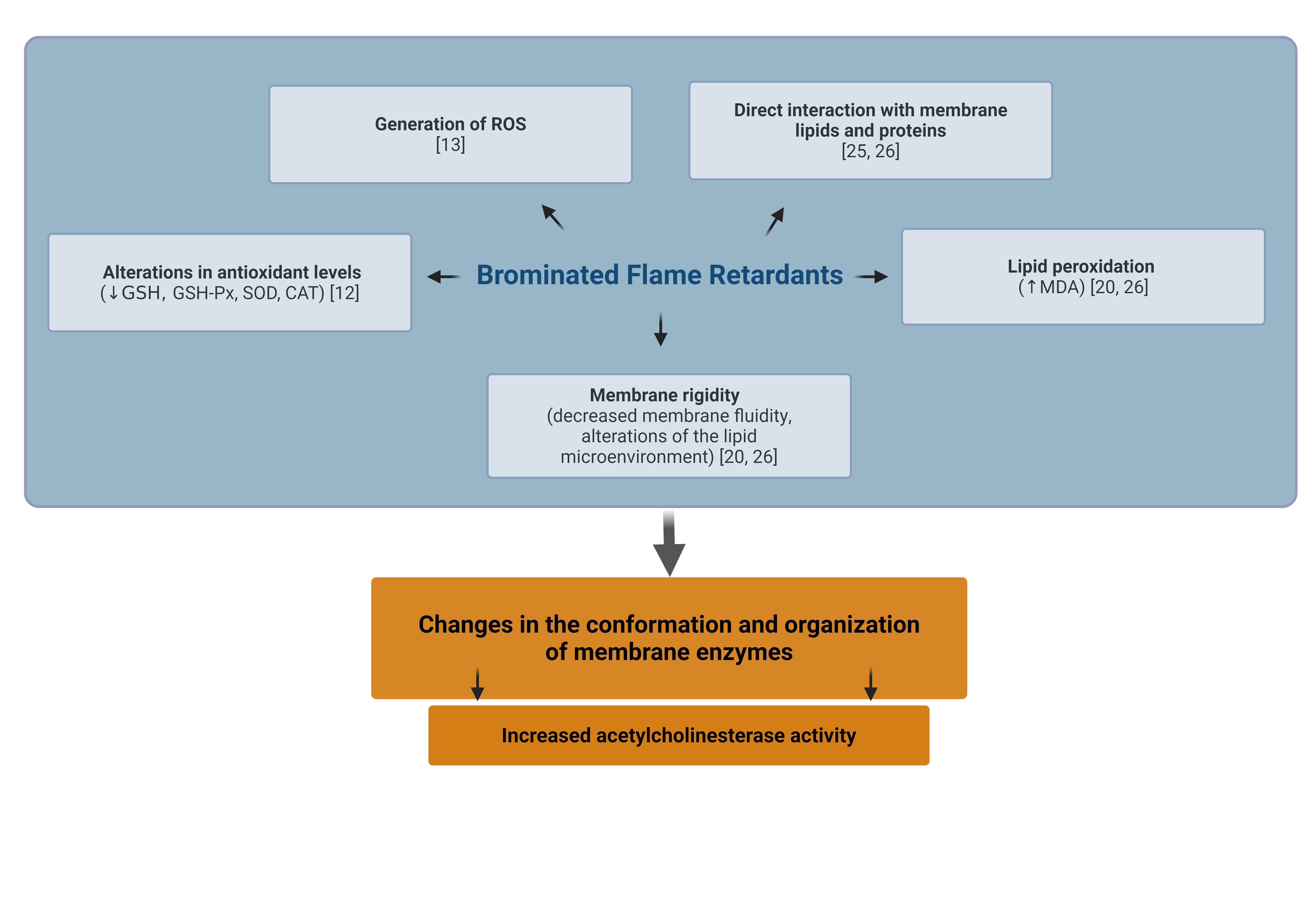
Fig. 3: Proposed Mechanisms of Bromophenol-Induced Upregulation of Acetylcholinesterase in Erythrocyte Membranes, according to previous studies [12, 13, 20, 25, 26]. Created in BioRender. Jarosiewicz, M. (2025) https://BioRender.com/kkjgkxi
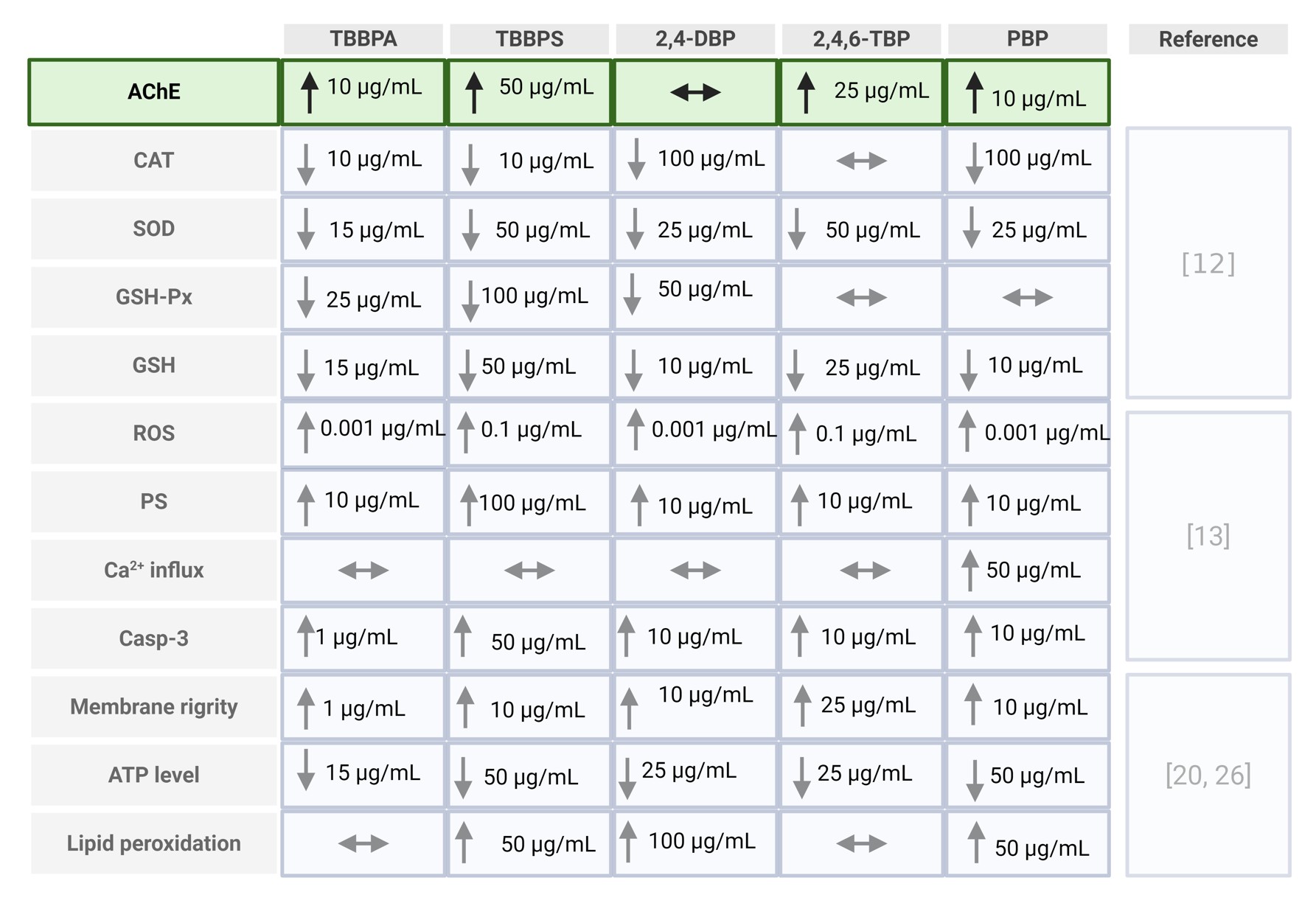
Table 1: Comparative summary of erythrocyte alterations induced by BFRs. Table integrates the newly obtained AChE data with erythrocyte parameters previously reported in independent publications [12, 13, 20, 25]. Created in BioRender. Jarosiewicz, M. (2026) https://BioRender.com/54tdm5q
Conclusion
The study demonstrated that the increase in acetylcholinesterase (AChE) activity in erythrocytes upon exposure to the tested BFRs may result from membrane structural alterations induced by oxidative stress, eryptosis initiation, and indirect modifications of the enzyme’s microenvironment. Notably, changes in AChE activity induced by TBBPA and PBP were observed at very low concentrations (10 µg/mL), indicating the relatively strong toxic effect of these compounds on this enzyme. These findings suggest that erythrocytic AChE may serve as a biomarker of exposure to environmental xenobiotics, including BFRs. However, it should be emphasized that this enzyme cannot be considered a highly sensitive biosensor, as alterations in its activity are observed at concentrations that simultaneously induce modifications in other cellular parameters, such as lipid peroxidation, phosphatidylserine externalization, and changes in membrane fluidity. The action of BFRs depends on their chemical structure and concentration, further underscoring the need for detailed studies on the molecular mechanisms underlying this interaction. Moreover, AChE may reflect not only the state of the cell membrane but also indicate early stages of eryptosis, increasing its potential diagnostic relevance. Further in vivo and in vitro studies are required to fully elucidate the effects of chronic BFR exposure on human health and its possible long-term biological consequences.
Acknowledgements
Author Contributions
Conceptualization: BB; methodology setup and optimization: BB, MJ; experimental work and analytical determinations: MJ; data interpretation: MM, BB; statistical analysis: MJ; data visualization and graphical preparation: MJ; manuscript preparation and writing: MJ, BB; critical review and supervision: BB. All authors read and approved the final manuscript.
Funding Sources
This work was funded by the statutory research admitted for Department of Biophysics of Environmental Pollution, University of Lodz and Grant for Young Researchers and PhD Students in 2016 Year funded by the University of Lodz (B1611000001167.02).
Statement of Ethics
The study protocol has been approved by the Bioethics Committee of the University of Lodz No. 7/KBBN-UŁ/II/2015.
Disclosure Statement
The authors have no conflicts of interest to declare.
References
- Zhang M, Buekens A, Li X: Brominated flame retardants and the formation of dioxins and furans in fires and combustion. J Hazard Mater 2016; 304, 26-39. https://doi.org/10.1016/j.jhazmat.2015.10.014
- Feiteiro J, Mariana M, Cairrão E: Health toxicity effects of brominated flame retardants: From environmental to human exposure. Environ Pollut 2021; 285, 117475. https://doi.org/10.1016/j.envpol.2021.117475
- EFSA Panel on Contaminants in the Food Chain (CONTAM): Schrenk D, Bignami M, Bodin L, Chipman JK, del Mazo J, et al.: Update of the scientific opinion on tetrabromobisphenol A (TBBPA) and its derivatives in food. EFSA Journal 2024, 22(7), e8859. https://doi.org/10.2903/j.efsa.2024.8859
- ECHA, 2022: https://echa.europa.eu/documents/10162/3dfab98b-2863-91bb-71a4-c9c0859d2342
- Zhang Y, Wang X, Chen C, An J, Shang Y, Li H, Xia H, Yu J, Wang C, Liu Y, Guo S. Regulation of TBBPA-induced oxidative stress on mitochondrial apoptosis in L02 cells through the Nrf2 signaling pathway. Chemosphere 2019; 226:463-71.https://doi.org/10.1016/j.chemosphere.2019.03.167
- Hamers T, Kamstra JH, Sonneveld E, Murk AJ, Kester MH, Andersson PL, Legler J, Brouwer A. In vitro profiling of the endocrine-disrupting potency of brominated flame retardants. Toxicol Sci 2006; 92(1):157-73. https://doi.org/10.1093/toxsci/kfj187
- Jagnytsch O, Opitz R, Lutz I, Kloas W. Effects of tetrabromobisphenol A on larval development and thyroid hormone-regulated biomarkers of the amphibian Xenopus laevis. Environ Res 2006; 101(3): 340-8. https://doi.org/10.1016/j.envres.2005.09.006
- Chen J, Tanguay RL, Simonich M, Nie S, Zhao Y, Li L, Bai C, Dong Q, Huang C, Lin K. TBBPA chronic exposure produces sex-specific neurobehavioral and social interaction changes in adult zebrafish. Neurotoxicol Teratol 2016; 56: 9-15. https://doi.org/10.1016/j.ntt.2016.05.008
- Yin N, Liang S, Liang S, Yang R, Hu B, Qin Z, Liu A, Faiola F. TBBPA and its alternatives disturb the early stages of neural development by interfering with the NOTCH and WNT pathways. Environ Sci Technol 2018; 52(9):5459-68. https://doi.org/10.1021/acs.est.8b00414
- Kibakaya EC, Stephen K, Whalen MM. Tetrabromobisphenol A has immunosuppressive effects on human natural killer cells. J Immunotoxicol 2009; 6(4): 285-92 https://doi.org/10.3109/15476910903258260
- Oral D, Balci A, Chao MW, Erkekoglu P: Toxic effects of tetrabromobisphenol A: focus on endocrine disruption. J Environ Pathol Toxicol Oncol 2021; 40(3). https://doi.org/10.1615/JEnvironPatholToxicolOncol.2021035595
- Jarosiewicz M, Krokosz A, Marczak A, Bukowska B: Changes in the activities of antioxidant enzymes and reduced glutathione level in human erythrocytes exposed to selected brominated flame retardants. Chemosphere 2019a; 227, 93-99. https://doi.org/10.1016/j.chemosphere.2019.04.008
- Jarosiewicz M, Michałowicz J, Bukowska B: In vitro assessment of eryptotic potential of tetrabromobisphenol A and other bromophenolic flame retardants. Chemosphere 2019b; 215, 404-412. https://doi.org/10.1016/j.chemosphere.2018.09.161
- Jarosiewicz M, Duchnowicz P, Włuka A, Bukowska B: Evaluation of the effect of brominated flame retardants on hemoglobin oxidation and hemolysis in human erythrocytes. Food Chem Toxicol 2017; 109, 264-271. https://doi.org/10.1016/j.fct.2017.09.016
- Ellman GL, Courtney KD, Andres Jr V, Featherstone RM: A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 1961; 7(2), 88-95. https://doi.org/10.1016/0006-2952(61)90145-9
- Lionetto MG, Caricato R, Calisi A, Giordano ME, Schettino T: Acetylcholinesterase as a biomarker in environmental and occupational medicine: new insights and future perspectives. Biomed Res Int 2013; (1), 321213. https://doi.org/10.1155/2013/321213
- Assis CR, Linhares AG, Cabrera MP, Oliveira VM, Silva KC, Marcuschi M, et al.: Erythrocyte acetylcholinesterase as biomarker of pesticide exposure: new and forgotten insights. Environm Sci Pollut Res 2018; 25(19), 18364-18376. https://doi.org/10.1007/s11356-018-2303-9
- Saldanha C: Human erythrocyte acetylcholinesterase in health and disease. Molecules 2017; 22(9), 1499. https://doi.org/10.3390/molecules22091499
- Bukowska B, Pieniazek D, Hutnik K, Duda W: Acetylo- i butyrylocholinoestreraza - budowa, funkcje i ich inhibitory, Curr Top Biophys 2007; 30: 11-23.
- Jarosiewicz M, Duchnowicz P, Jarosiewicz P, Huras B, Bukowska B: An in vitro comparative study of the effects of tetrabromobisphenol A and tetrabromobisphenol S on human erythrocyte membranes - changes in ATP level, perturbations in membrane fluidity, alterations in conformational state and damage to proteins. Int J Mol Sci 2021; 22(17), 9443. https://doi.org/10.3390/ijms22179443
- Maćczak A, Duchnowicz P, Sicińska P, Koter-Michalak M, Bukowska B, Michałowicz J: The in vitro comparative study of the effect of BPA, BPS, BPF and BPAF on human erythrocyte membrane; perturbations in membrane fluidity, alterations in conformational state and damage to proteins, changes in ATP level and Na+/K+ ATPase and AChE activities. Food ChemToxicol 2017; 110, 351-359. https://doi.org/10.1016/j.fct.2017.10.028
- Copeto S, Ganço S, Ferreira IJ, Silva M, Motta C, Diniz M: The Effects of Tetrabromobisphenol A (TBBPA) on the Mussel Mytilus galloprovincialis: A Multi-Biomarker Approach. Oceans 2024; 5(2), 181-195. https://doi.org/10.3390/oceans5020011
- Wang J, Dai GD: Comparative effects of brominated flame retardants BDE-209, TBBPA, and HBCD on neurotoxicity in mice. Chem Res Toxicol 2022; 35(9), 1512-1518. https://doi.org/10.1021/acs.chemrestox.2c00126
- Naciff JM, Khambatta ZS, Reichling TD, Carr GJ, Tiesman JP, Singleton DW, et al. The genomic response of Ishikawa cells to bisphenol A exposure is dose- and time-dependent. Toxicology. 2010;270(2-3):137-149. https://doi.org/10.1016/j.tox.2010.02.008
- Jarosiewicz M, Miłowska K, Krokosz A, Bukowska B: Evaluation of the effect of selected brominated flame retardants on human serum albumin and human erythrocyte membrane proteins. Int J Mol Sci 2020; 21(11), 3926. https://doi.org/10.3390/ijms21113926
- Jarosiewicz M, Duchnowicz P, Bukowska B: Assessment of membrane and metabolic parameters of erythrocytes exposed to selected bromophenols. Sci Rep 2025; 15(1), 13004. https://doi.org/10.1038/s41598-025-97707-8
- Carvalho FA, de Almeida JL, Freitas-Santos T, Saldanha C: Modulation of erythrocyte acetylcholinesterase activity and its association with G protein-band 3 interactions. J Membr Biol 2009; 228(2), 89-97. https://doi.org/10.1007/s00232-009-9162-8
- Franco RS, Puchulu-Campanella ME, Barber LA, Palascak MB, Joiner CH, Low PS, Cohen RM: Changes in the properties of normal human red blood cells during in vivo aging. Am J Hematol 2013; 88, 44-51. https://doi.org/10.1002/ajh.23344
- Leal JKF, Adjobo-Hermans MJ, Brock, Bosman GJ: Acetylcholinesterase provides new insights into red blood cell ageing in vivo and in vitro. J Blood Transfus 2017; 15(3), 232.